Always read the methods section: Formaldehyde emissions
By Brent Stephens on May 3, 2012
I posted a link on Twitter the other day (April 30) about a study investigating the inhalation exposure to formaldehyde from the use of personal care products (i.e., lotions, conditioners, shampoos, etc.). Basically, the study involved using a relatively large amount of several products in a simulated bathroom and measuring the resulting concentrations of formaldehyde. In their interpretation of their results, they said:
“Overall, our data yielded evidence that inhalation of FA [formaldehyde] from the use of PCP [personal care products] containing FA-releasers poses no risk to human health.”
I posted the link, then thought about the study a day later, wondering about its methods. So I dug a little deeper, and found the methods and results to be somewhat misleading, depending on your point of view.
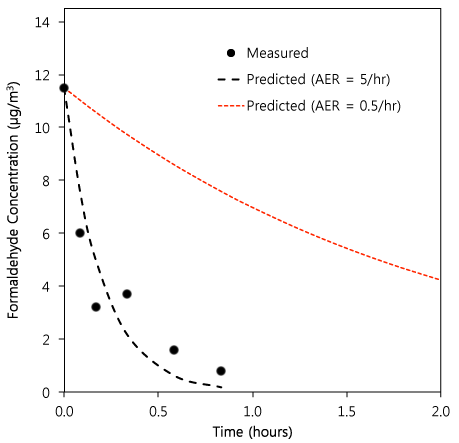
In their results, they reported both peak and 1-hour-mean formaldehyde concentrations. In order to explore what those really mean, let’s take a look at the highest emitter: body lotion. They applied nearly 8 grams of this stuff and measured the subsequent formaldehyde concentration in their bathroom. A peak of about 11.5 µg per cubic meters was reached with a 1-hour-mean room concentration of about half that, decaying below baseline within 45 minutes or so. So they can definitely claim low 1-hour averages in this setup.
Sounds reasonable, right? Well, it all depends on what you think of their experimental setup. Dig a little deeper into the methods section and you see their bathroom had a volume of 9.4 cubic meters with an air exchange rate of approximately 5 per hour. That’s a large air exchange rate, likely representative of a situation with an exhaust fan running. Natural air exchange rates (AER) in US residences are much lower, on average, when fans aren’t operating (i.e., 0.4-0.5 per hour on average). Let’s take an AER of 0.5 per hour, and look at the likely difference in the time for the indoor concentration to decay:
Figure 1. Dots represent measured values from the study. The dashed black line represents a simple first-order model of the concentration decay from the initial peak value [C(t) = exp(-AER*time), where AER = 5/hr]. The dashed red line represents the same decay model with AER = 0.5/hr.
Naturally, the AER makes a big difference in the 1-hour mean values. One could argue that if you’re trying to replicate a ‘worst-case’ environment (they do make this argument), you would want to control the amount that is applied (which they did; they applied a large amount of lotion) as well as the AER (which they did not set conservatively; they elevated it an order of magnitude higher than a typical residence without an exhaust fan operating). So they might be overstating the safety of these products. On the other hand, if you look at ASHRAE Standard 62.2, which provides IAQ guidelines for residences, they suggest bathroom exhaust fans should provide 50 CFM (or 25 liters per second of flow). That’s about 90 cubic meters per hour, or an air exchange rate of about 9.6 per hour (90 cubic meters per hour divided by 9.4 cubic meters). So you could also make the argument in the other direction; that is, their data were actually conservative for a US residence with guideline values of bathroom exhaust flow rates.
So I think the take home message is twofold: (1) always pay attention to experimental methods, and (2) in studies like these, you have to be very careful and open with your experimental setup. Emission studies are generally more helpful if they report actual emission rates (in units of mass of pollutant per time) or emission factors (in units of mass of pollutant emitted per mass of product used). Otherwise, your results end up being largely a function of the environment in which it was performed, which diminishes the relevance in other situations.
Filed under: Experimental methods